Reading Esential for Physical Science Chapert 18 Chemical Bons Nswer Key
Hydrogen Bonding
- Folio ID
- 1660
A hydrogen bond is an intermolecular force (Imf) that forms a special type of dipole-dipole attraction when a hydrogen atom bonded to a strongly electronegative atom exists in the vicinity of another electronegative atom with a lone pair of electrons. Intermolecular forces (IMFs) occur between molecules. Other examples include ordinary dipole-dipole interactions and dispersion forces. Hydrogen bonds are are mostly stronger than ordinary dipole-dipole and dispersion forces, only weaker than true covalent and ionic bonds.
The evidence for hydrogen bonding
Many elements class compounds with hydrogen. If you plot the boiling points of the compounds of the group 14 elements with hydrogen, you notice that the boiling points increase as you go downwards the group.
The increase in humid point happens considering the molecules are getting larger with more than electrons, so van der Waals dispersion forces get greater. If you echo this do with the compounds of the elements in groups fifteen, 16, and 17 with hydrogen, something odd happens.
Although the same reasoning applies for group 4 of the periodic tabular array, the boiling signal of the chemical compound of hydrogen with the first element in each group is abnormally high. In the cases of \(NH_3\), \(H_2O\) and \(HF\) there must be some boosted intermolecular forces of attraction, requiring significantly more than heat energy to suspension the IMFs. These relatively powerful intermolecular forces are described as hydrogen bonds.
Origin of Hydrogen Bonding
The molecules capable of hydrogen bonding include the following:
Notice that in each of these molecules:
- The hydrogen is fastened directly to a highly electronegative atoms, causing the hydrogen to larn a highly positive charge.
- Each of the highly electronegative atoms attains a high negative charge and has at least one "active" lone pair. Solitary pairs at the 2-level accept electrons independent in a relatively small volume of space, resulting in a high negative charge density. Solitary pairs at college levels are more lengthened and, resulting in a lower charge density and lower affinity for positive charge.
If you lot are non familiar with electronegativity, you should follow this link before you continue.
Consider two water molecules coming close together.
The \(\delta^+\) hydrogen is so strongly attracted to the lone pair that it is nearly as if yous were start to form a co-ordinate (dative covalent) bail. It doesn't get that far, but the allure is significantly stronger than an ordinary dipole-dipole interaction. Hydrogen bonds have well-nigh a tenth of the strength of an average covalent bond, and are constantly broken and reformed in liquid water. If you lot liken the covalent bond between the oxygen and hydrogen to a stable marriage, the hydrogen bond has "but good friends" status.
H2o is an ideal example of hydrogen bonding. Find that each h2o molecule can potentially form four hydrogen bonds with surrounding h2o molecules: two with the hydrogen atoms and two with the with the oxygen atoms. There are exactly the right numbers of \(\delta^+\) hydrogens and lone pairs for every one of them to be involved in hydrogen bonding.
This is why the boiling point of water is higher than that of ammonia or hydrogen fluoride. In the case of ammonia, the corporeality of hydrogen bonding is express by the fact that each nitrogen only has one lonely pair. In a group of ammonia molecules, there are not enough alone pairs to go around to satisfy all the hydrogens. In hydrogen fluoride, the problem is a shortage of hydrogens. In water, two hydrogen bonds and two lone pairs permit formation of hydrogen bond interactions in a lattice of water molecules. Water is thus considered an platonic hydrogen bonded organization.
More than complex examples of hydrogen bonding
The hydration of negative ions
When an ionic substance dissolves in water, h2o molecules cluster around the separated ions. This process is chosen hydration. Water frequently attaches to positive ions by according (dative covalent) bonds. It bonds to negative ions using hydrogen bonds.
If you are interested in the bonding in hydrated positive ions, you could follow this link to co-ordinate (dative covalent) bonding.
The diagram shows the potential hydrogen bonds formed with a chloride ion, Cl-. Although the lonely pairs in the chloride ion are at the 3-level and would non normally be active enough to form hydrogen bonds, they are made more than bonny by the full negative charge on the chlorine in this case.
All the same complicated the negative ion, there will always be lone pairs that the hydrogen atoms from the water molecules can hydrogen bond to.
Hydrogen bonding in alcohols
An alcohol is an organic molecule containing an -OH grouping. Any molecule which has a hydrogen cantlet fastened directly to an oxygen or a nitrogen is capable of hydrogen bonding. Hydrogen bonds also occur when hydrogen is bonded to fluorine, simply the HF grouping does not appear in other molecules. Molecules with hydrogen bonds will always take higher boiling points than similarly sized molecules which don't have an an -O-H or an -N-H grouping. The hydrogen bonding makes the molecules "stickier," such that more than heat (energy) is required to separate them. This phenomenon can be used to analyze boiling point of different molecules, defined as the temperate at which a phase change from liquid to gas occurs.
Ethanol, \(\ce{CH3CH2-O-H}\), and methoxymethane, \(\ce{CH3-O-CH3}\), both have the same molecular formula, \(\ce{C2H6O}\).
They have the same number of electrons, and a similar length. The van der Waals attractions (both dispersion forces and dipole-dipole attractions) in each will be like. However, ethanol has a hydrogen cantlet attached direct to an oxygen; here the oxygen nonetheless has two alone pairs like a water molecule. Hydrogen bonding can occur between ethanol molecules, although not equally effectively as in water. The hydrogen bonding is limited by the fact that there is but one hydrogen in each ethanol molecule with sufficient + charge.
In methoxymethane, the lonely pairs on the oxygen are withal there, simply the hydrogens are not sufficiently + for hydrogen bonds to form. Except in some rather unusual cases, the hydrogen cantlet has to be fastened straight to the very electronegative element for hydrogen bonding to occur. The humid points of ethanol and methoxymethane evidence the dramatic effect that the hydrogen bonding has on the stickiness of the ethanol molecules:
| ethanol (with hydrogen bonding) | 78.5°C |
| methoxymethane (without hydrogen bonding) | -24.8°C |
The hydrogen bonding in the ethanol has lifted its boiling point nigh 100°C. It is important to realize that hydrogen bonding exists in improver to van der Waals attractions. For example, all the following molecules contain the same number of electrons, and the first two have like chain lengths. The higher boiling point of the butan-1-ol is due to the additional hydrogen bonding.
Comparing the two alcohols (containing -OH groups), both boiling points are high because of the additional hydrogen bonding; however, the values are non the aforementioned. The humid point of the 2-methylpropan-1-ol isn't equally high as the butan-one-ol because the branching in the molecule makes the van der Waals attractions less effective than in the longer butan-1-ol.
Hydrogen bonding in organic molecules containing nitrogen
Hydrogen bonding too occurs in organic molecules containing North-H groups; call back the hydrogen bonds that occur with ammonia. Examples range from simple molecules like CH3NHtwo (methylamine) to large molecules like proteins and Deoxyribonucleic acid. The two strands of the famous double helix in DNA are held together by hydrogen bonds between hydrogen atoms fastened to nitrogen on i strand, and lone pairs on another nitrogen or an oxygen on the other one.
Donors and Acceptors
In order for a hydrogen bond to occur there must exist both a hydrogen donor and an acceptor present. The donor in a hydrogen bond is commonly a strongly electronegative atom such as N, O, or F that is covalently bonded to a hydrogen bail.
The hydrogen acceptor is an electronegative cantlet of a neighboring molecule or ion that contains a lone pair that participates in the hydrogen bail.
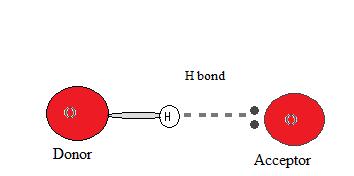
Why does a hydrogen bond occur?
Since the hydrogen donor (N, O, or F) is strongly electronegative, information technology pulls the covalently bonded electron pair closer to its nucleus, and abroad from the hydrogen atom. The hydrogen atom is then left with a partial positive charge, creating a dipole-dipole attraction betwixt the hydrogen atom bonded to the donor and the lone electron pair of the acceptor. This results in a hydrogen bond.(see Interactions Betwixt Molecules With Permanent Dipoles)
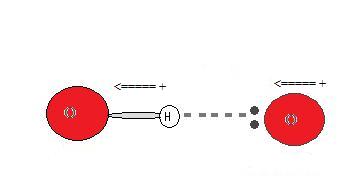
Types of hydrogen bonds
Although hydrogen bonds are well-known as a type of IMF, these bonds can as well occur within a single molecule, between 2 identical molecules, or between two dissimilar molecules.
Intramolecular hydrogen bonds
Intramolecular hydrogen bonds are those which occur inside one single molecule. This occurs when 2 functional groups of a molecule tin can class hydrogen bonds with each other. In order for this to happen, both a hydrogen donor a hydrogen acceptor must be present inside i molecule, and they must be within close proximity of each other in the molecule. For example, intramolecular hydrogen bonding occurs in ethylene glycol (C2H4(OH)2) between its two hydroxyl groups due to the molecular geometry.
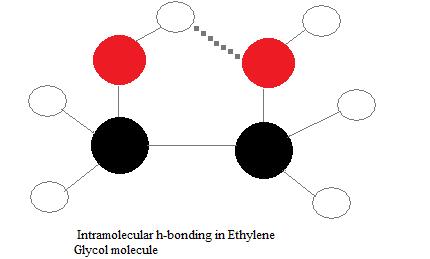
Intermolecular hydrogen bonds
Intermolecular hydrogen bonds occur between dissever molecules in a substance. They can occur betwixt any number of like or different molecules as long as hydrogen donors and acceptors are nowadays in positions where they can interact with one some other. For example, intermolecular hydrogen bonds can occur between NHiii molecules lonely, between H2O molecules alone, or between NH3 and H2O molecules.
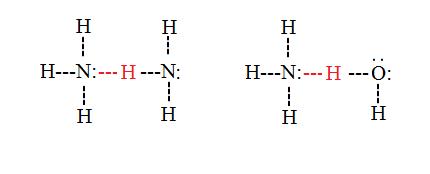
Properties and effects of hydrogen bonds
On Boiling Point
When we consider the boiling points of molecules, we usually look molecules with larger tooth masses to have higher normal boiling points than molecules with smaller tooth masses. This, without taking hydrogen bonds into business relationship, is due to greater dispersion forces (see Interactions Betwixt Nonpolar Molecules). Larger molecules have more space for electron distribution and thus more possibilities for an instantaneous dipole moment. Notwithstanding, when we consider the tabular array below, we run into that this is not always the instance.
| Chemical compound | Molar Mass | Normal Boiling Indicate |
|---|---|---|
| \(H_2O\) | 18 grand/mol | 373 K |
| \(HF\) | xx g/mol | 292.5 K |
| \(NH_3\) | 17 g/mol | 239.8 Thousand |
| \(H_2S\) | 34 thousand/mol | 212.9 K |
| \(HCl\) | 36.4 g/mol | 197.9 Thou |
| \(PH_3\) | 34 g/mol | 185.two K |
We come across that H2O, HF, and NHthree each accept higher boiling points than the same chemical compound formed between hydrogen and the next chemical element moving down its respective group, indicating that the erstwhile have greater intermolecular forces. This is because HiiO, HF, and NHiii all exhibit hydrogen bonding, whereas the others do not. Furthermore, \(H_2O\) has a smaller tooth mass than HF simply partakes in more than hydrogen bonds per molecule, so its boiling indicate is college.
On Viscosity
The same outcome that is seen on boiling point as a result of hydrogen bonding can also be observed in the viscosity of certain substances. Substances capable of forming hydrogen bonds tend to have a higher viscosity than those that practice not for hydrogen bonds. Generally, substances that have the possibility for multiple hydrogen bonds showroom even higher viscosities.
Factors preventing Hydrogen bonding
Electronegativity
Hydrogen bonding cannot occur without significant electronegativity differences between hydrogen and the atom it is bonded to. Thus, we run into molecules such as PH3, which no not partake in hydrogen bonding. PH3 exhibits a trigonal pyramidal molecular geometry similar that of ammonia, merely unlike NHiii it cannot hydrogen bail. This is due to the similarity in the electronegativities of phosphorous and hydrogen. Both atoms have an electronegativity of 2.1, and thus, no dipole moment occurs. This prevents the hydrogen bonding from acquiring the partial positive charge needed to hydrogen bond with the lone electron pair in some other molecule. (meet Polarizability)
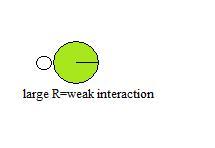
Atom Size
The size of donors and acceptors can as well effect the power to hydrogen bond. This can business relationship for the relatively low ability of Cl to form hydrogen bonds. When the radii of two atoms differ greatly or are big, their nuclei cannot achieve close proximity when they collaborate, resulting in a weak interaction.
Hydrogen Bonding in Nature
Hydrogen bonding plays a crucial role in many biological processes and can account for many natural phenomena such as the Unusual properties of H2o. In addition to being nowadays in water, hydrogen bonding is likewise important in the water send arrangement of plants, secondary and third protein structure, and Dna base pairing.
Plants
The cohesion-adhesion theory of transport in vascular plants uses hydrogen bonding to explain many key components of h2o movement through the plant's xylem and other vessels. Within a vessel, water molecules hydrogen bail not only to each other, just also to the cellulose chain which comprises the wall of plant cells. Since the vessel is relatively modest, the attraction of the water to the cellulose wall creates a sort of capillary tube that allows for capillary activity. This mechanism allows plants to pull water upward into their roots. Furthermore, hydrogen bonding tin can create a long chain of water molecules which tin can overcome the force of gravity and travel up to the loftier altitudes of leaves.
Proteins
Hydrogen bonding is present abundantly in the secondary construction of proteins, and as well sparingly in third conformation. The secondary structure of a protein involves interactions (mainly hydrogen bonds) between neighboring polypeptide backbones which contain Nitrogen-Hydrogen bonded pairs and oxygen atoms. Since both Northward and O are strongly electronegative, the hydrogen atoms bonded to nitrogen in one polypeptide courage can hydrogen bond to the oxygen atoms in another concatenation and visa-versa. Though they are relatively weak, these bonds offer substantial stability to secondary protein construction considering they repeat many times and piece of work collectively.
In tertiary protein structure, interactions are primarily between functional R groups of a polypeptide concatenation; one such interaction is called a hydrophobic interaction. These interactions occur because of hydrogen bonding betwixt water molecules effectually the hydrophobe that further reinforces protein conformation.
References
- Brown, et al. Chemistry:The Fundamental Science. 11th ed. Upper Saddle River, New Jersey: Pearson/Prentice Hall, 2008.
- Chang, Raymond. Full general Chemical science:The Essential Concepts. third ed. New York: Mcgraw Hill, 2003
- Petrucci, et al. General Chemistry: Principles & Modernistic Applications. ninth ed. Upper Saddle River, New Bailiwick of jersey: Pearson/Prentice Hall, 2007.
Source: https://chem.libretexts.org/Bookshelves/Physical_and_Theoretical_Chemistry_Textbook_Maps/Supplemental_Modules_(Physical_and_Theoretical_Chemistry)/Physical_Properties_of_Matter/Atomic_and_Molecular_Properties/Intermolecular_Forces/Specific_Interactions/Hydrogen_Bonding
0 Response to "Reading Esential for Physical Science Chapert 18 Chemical Bons Nswer Key"
Post a Comment